Chapter 8: Q. 8.47 (page 276)
What happens to the volume of a bicycle tire or a basketball when you use an air pump to add air?
Short Answer
The volume of a bicycle tire or a basketball when adding the air by a pump then volume is increases.
Learning Materials
Features
Discover
Chapter 8: Q. 8.47 (page 276)
What happens to the volume of a bicycle tire or a basketball when you use an air pump to add air?
The volume of a bicycle tire or a basketball when adding the air by a pump then volume is increases.
All the tools & learning materials you need for study success - in one app.
Get started for free
Use the molar volume to calculate each of the following at STP:
a. the number of moles of O2in 44.8 L of O2 gas.
b. the volume in liters, occupied by 2.50 moles of N2 gas.
c. the volume, in liters, occupied by 50.0 g of Ar gas.
d. the number of grams of H2 in 16.20 mL of H2 gas.
A gas mixture containing oxygen, nitrogen, and neon exerts a total pressure of . If helium added to the mixture increases the pressure to , what is the partial pressure, in atmospheres, of the helium?
A balloon is filled with helium gas at a partial pressure of and neon gas with a partial pressure of . For each of the following changes (a to e) of the initial balloon, select the diagram (A, B, or C) that shows the final volume of the balloon:
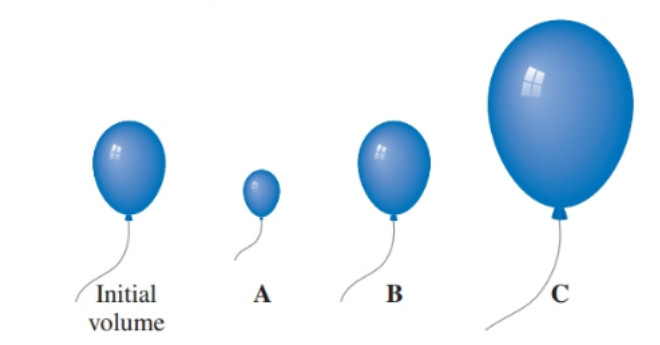
a. The balloon is put in a cold storage unit (do not change).
b. The balloon floats to a higher altitude where the pressure is less (do not change).
c. All of the neon gas is removed (do not change).
d. The Kelvin temperature doubles and half of the gas atoms leak out (does not change).
e.gas is added (do not change).
A sample of neon initially has a volume of at . What final temperature, in degrees Celsius, is needed to change the volume of the gas to each of the following, if and do not change?
a.
b .
c.
d.
A gas sample has a volume of with an unknown temperature. The same gas has a volume of when the temperature is , with no change in the pressure or amount of gas. What was the initial temperature, in degrees Celsius, of the gas?
What do you think about this solution?
We value your feedback to improve our textbook solutions.