Chapter 8: Q. 8.39 (page 271)
A tank contains isoflurane, an inhaled anesthetic, at a pressure of and . What is the pressure, in atmospheres, if the gas is warmed to a temperature of, ifdo not change?
Short Answer
The final pressure of the gas is.
Learning Materials
Features
Discover
Chapter 8: Q. 8.39 (page 271)
A tank contains isoflurane, an inhaled anesthetic, at a pressure of and . What is the pressure, in atmospheres, if the gas is warmed to a temperature of, ifdo not change?
The final pressure of the gas is.
All the tools & learning materials you need for study success - in one app.
Get started for free
The air in a cylinder with a piston has a volume of mL and
a pressure of mmHg.
a.To obtain a higher pressure inside the cylinder at constant
temperature and amount of gas, would the cylinder change
as shown in A or B? Explain your choice.
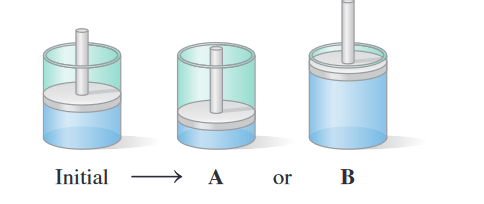
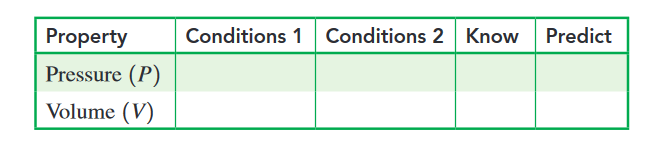
b. If the pressure inside the cylinder increases to atm, what
is the final volume, in milliliters, of the cylinder? Complete
the following data table
A fire extinguisher has a pressure of at . What is the final pressure, in atmospheres, when the fire extinguisher is used at a temperature of , if do not change?
A gas sample has a pressure of when the temperature is . What is the final pressure, in atmospheres, when the temperature is , with no change in the volume or amount of gas?
A sample of Ne gas has a pressure of 654 mmHg with an unknown volume. The gas has a pressure of 345 mmHg when the volume is 495 mL, with no change in temperature or amount of gas. What was the initial volume, in milliliters, of the gas?
On a climb up Mt. Whitney, the atmospheric pressure drops to mmHg. What is the pressure in terms of the following units?
a. atm
b. torr
What do you think about this solution?
We value your feedback to improve our textbook solutions.