Chapter 2: Q.2.107 (page 57)
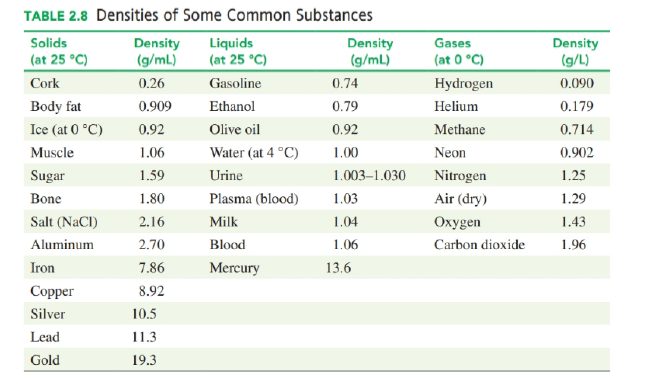
How many millilitres of gasoline has a mass of 1.2 kg (see Table 2.8)? (2.7)
Short Answer
ml
Learning Materials
Features
Discover
Chapter 2: Q.2.107 (page 57)
How many millilitres of gasoline has a mass of 1.2 kg (see Table 2.8)? (2.7)
ml
All the tools & learning materials you need for study success - in one app.
Get started for free
Use the density values in Table 2.8 to solve each of the following problems:
a. What is the mass, in grams, of a cube of copper that has a volume of 74.1 cm3?
b. How many kilograms of gasoline fill a 12.0-gal gas tank?
c. What is the volume, in cubic centimeters, of an ice cube that has a mass of 27 g?
The price of of potatoes is . If all the potatoes sold today at the store bring in , how many kilograms of potatoes did grocery shoppers buy? (2.6)
How can you check that you have written the correct conversion factors for an equality.
State the type of measurement in each of the following statements:
a. I put of gasoline in my gas tank.
b. My friend is tall.
c. Earth is away from the Moon.
d. The horse won the race by .
a. Write an equality and two conversion factors for Greg's serum iron level.
b. How many micrograms of iron were in the 8.0-ml Sample of Greg's blood?
What do you think about this solution?
We value your feedback to improve our textbook solutions.