Chapter 2: Q. 2.119 (page 56)
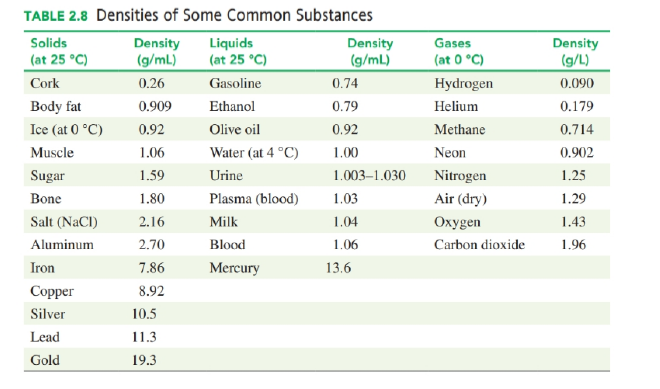
How many milliliters of olive oil has the same mass as of gasoline (see Table 2.8) ? (2,7)
Short Answer
Olive oil has the same mass as gasoline and has a volume of .
Learning Materials
Features
Discover
Chapter 2: Q. 2.119 (page 56)
How many milliliters of olive oil has the same mass as of gasoline (see Table 2.8) ? (2,7)
Olive oil has the same mass as gasoline and has a volume of .
All the tools & learning materials you need for study success - in one app.
Get started for free
A car travels at and gets of gasoline. How many gallons of gasoline are needed for a -h trip? (2,6)
Write the complete name for each of the following units:
a.
b.
c.
d.
Solve each of the following problems:
a. A glucose solution has a density of 1.02 g/mL. What is its specific gravity?
b. A 0.200 mL sample of very-low-density lipoprotein (VLDL) has a mass of 190 mg. What is the density of the VLDL?
c. Butter has a specific gravity of 0.86. What is the mass, in grams, of 2.15 L of butter?
d. A 5.000-mL urine sample has a mass of 5.025 g. If the normal range for the specific gravity of urine is 1.003 to 1.030, would the specific gravity of this urine sample indicate that the patient could have type 2 diabetes?
Use the density values in Table 2.8 to solve each of the following problems:
a. How many liters of ethanol contains 1.50 kg of ethanol?
b. How many grams of mercury are present in a barometer that holds 6.5 mL of mercury?
c. A sculptor has prepared a mold for casting a silver figure. The figure has a volume of 225 cm3. How many ounces of silver are needed in the preparation of the silver figure?
Write an equality and two conversion factors for each of the following medications:
(a) mg of Atarax per mL of Atarax syrup
(b) g of Lanoxin per tablet of Lanoxin
(c) mg of Motrin per tablet of Motrin
What do you think about this solution?
We value your feedback to improve our textbook solutions.