Chapter 7: Q. 7.6 (page 220)
Calculate each of the following quantities in moles of :
- moles of
- moles of
- atoms of
- atoms of
Short Answer
Quantities present in moles of are:
- moles of carbon
- moles of oxygen
- atoms of hydrogen
- atoms of carbon
Learning Materials
Features
Discover
Chapter 7: Q. 7.6 (page 220)
Calculate each of the following quantities in moles of :
Quantities present in moles of are:
All the tools & learning materials you need for study success - in one app.
Get started for free
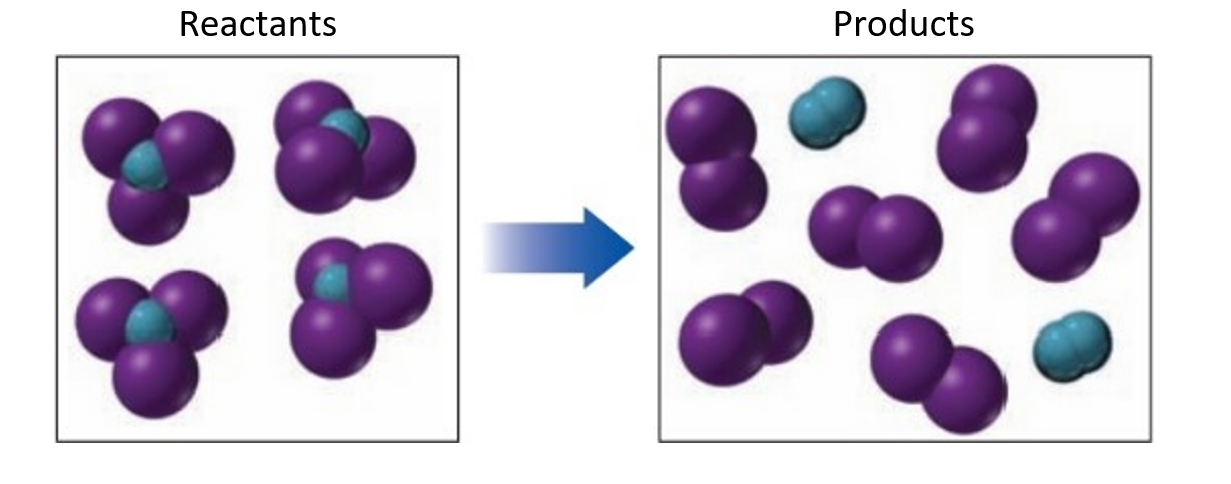
If blue spheres represent nitrogen atoms, purple spheres represent iodine atoms and the reacting molecules are solid, while the products are gases.
a. The compound , Epsom salts, is used to soothe sore feet and muscles. How many grams will you need to prepare a bath containing localid="1653460710134" mole of Epsom salts?
b. Potassium iodide, , is used as an expectorant. How many grams are inlocalid="1653460784056" moleof potassium iodide?
How many grams are in moleof each of the following? ()
a.
b.
c.
Identify each of the following as an oxidation or a reduction:
a.
b.
c.
d.
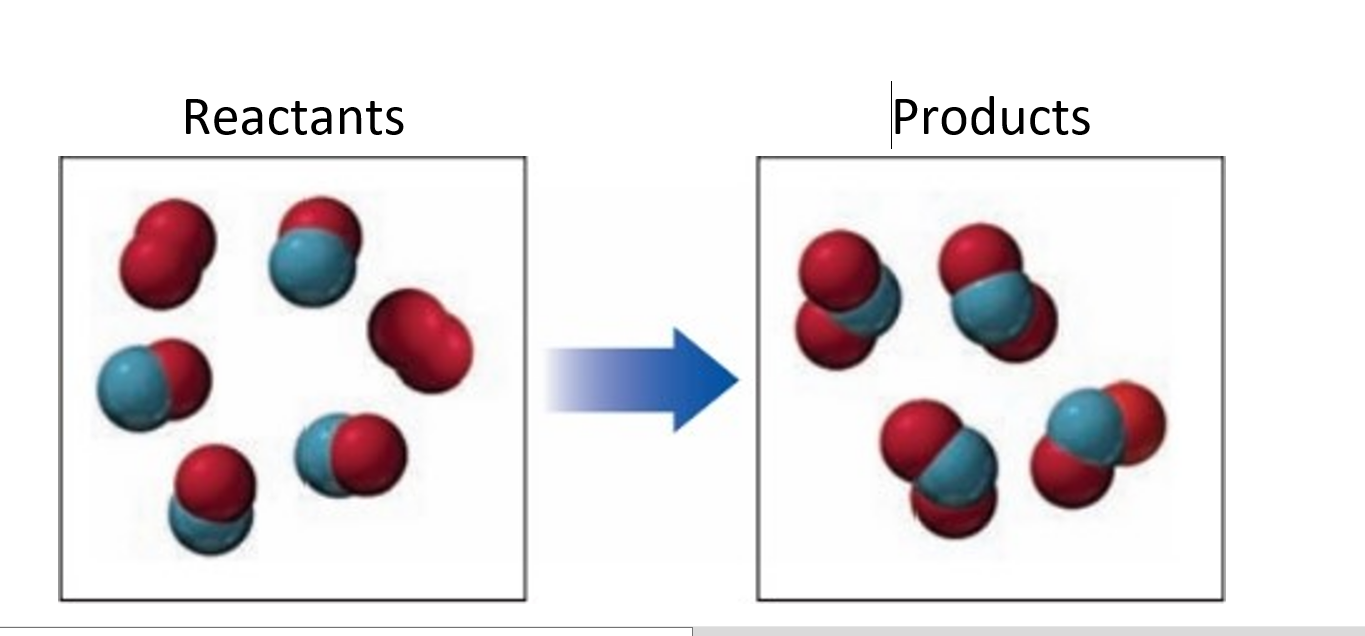
If red spheres represent oxygen atoms, blue spheres represent nitrogen atoms, and all the Molecules are gases,
What do you think about this solution?
We value your feedback to improve our textbook solutions.