Chapter 7: Q. 7.103 (page 255)
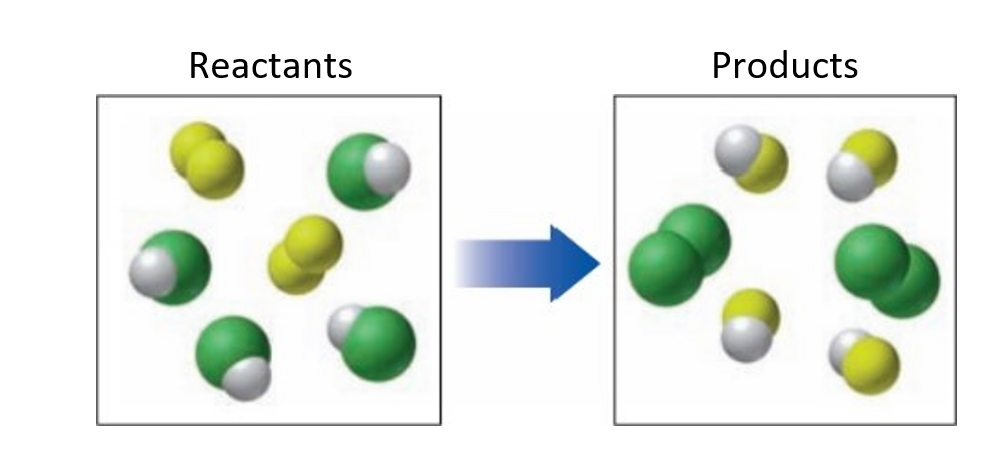
When ammonia () gas reacts with fluorine gas, the gaseous products are dinitrogen tetrafluoride () and hydrogen fluoride (). ()
a. Write the balanced chemical equation.
b. How many moles of each reactant are needed to produce localid="1653469855463" moleof ?
c. How many grams of are needed to react with localid="1653469902894" gof ?
d. How many grams of can be produced when localid="1653469925977" gof reacts?
Short Answer
(a) The required balanced equation is
(b) We require localid="1653470096757" moleof and localid="1653470331840" moleof
(c) The gof are needed to react with gof
(d) The gof are needed to react with gof