Chapter 14: Q. 14.2 (page 473)
What carboxylic acid is found in vinegar?
Short Answer
Acetate is the outcome of alcohol hydrolysis in alcohol and produce.
Learning Materials
Features
Discover
Chapter 14: Q. 14.2 (page 473)
What carboxylic acid is found in vinegar?
Acetate is the outcome of alcohol hydrolysis in alcohol and produce.
All the tools & learning materials you need for study success - in one app.
Get started for free
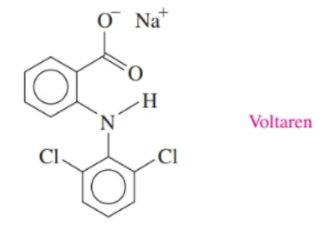
Voltaren is indicated for acute and chronic treatment of the symptoms of rheumatoid arthritis. Identify the functional groups in Voltaren.
What flavor would you notice if you smelled or tasted the following?
a. ethyl butanoate
b. propyl acetate
c. pentyl acetate
14.21 Draw the condensed structural formulas for and and line-angle formulas for and :
a. propyl butyrate
b. butyl formate
c. ethyl pentanoate
d. methyl propanoate
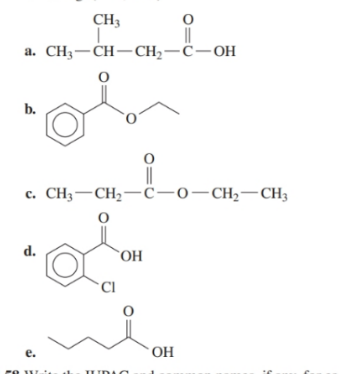
Write the IUPAC and common names, if any, for each of the following:
There are four amine isomers with the molecular formula . Draw their condensed structural formulas, write the common name, and classify each as a primary , secondary , or tertiary amine. (14.5)
What do you think about this solution?
We value your feedback to improve our textbook solutions.