Chapter 14: Q. 14.12 (page 477)
Write the balanced chemical equation for the reaction of each of the following carboxylic acids with :
a. acetic acid
b. 2-methylbutanoic acid
c. 4-chlorobenzoic acid
Short Answer
(part a) 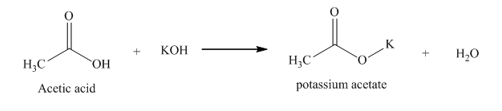
(part b) 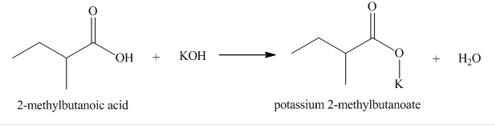
(part c) 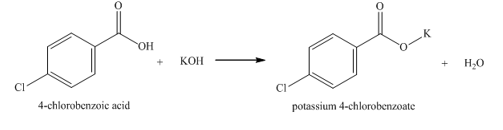
Learning Materials
Features
Discover
Chapter 14: Q. 14.12 (page 477)
Write the balanced chemical equation for the reaction of each of the following carboxylic acids with :
a. acetic acid
b. 2-methylbutanoic acid
c. 4-chlorobenzoic acid
(part a) 
(part b) 
(part c) 
All the tools & learning materials you need for study success - in one app.
Get started for free
What flavor would you notice if you smelled or tasted the following?
a. ethyl butanoate
b. propyl acetate
c. pentyl acetate
What carboxylic acid is responsible for the pain of an ant sting?
Write the common name for each of the following:
a. 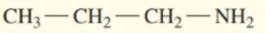
b. 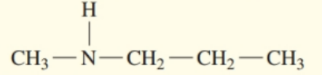
c. 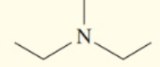
a. Identify the functional groups in dicyclanil.
b. The recommended application for dicyclanil for an adult sheep is 65 mg / kg of body mass. If dicyclanil is supplied in a spray with a concentration of 50.mg/mL, how many milliliters of the spray are required to treat a 70.-kg adult sheep?
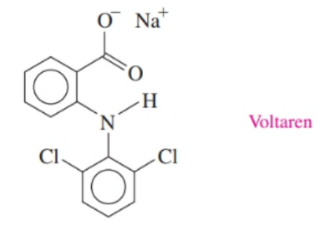
Voltaren is indicated for acute and chronic treatment of the symptoms of rheumatoid arthritis. Identify the functional groups in Voltaren.
What do you think about this solution?
We value your feedback to improve our textbook solutions.