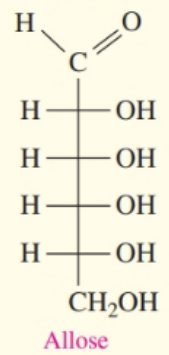
Chapter 13: Q. 13.70 (page 467)
Use the Fischer projection for D-gulose in problem to answer each of the following:
a. Draw the Fischer projection and name the product formed by the reduction of D-gulose.
b. Draw the Fischer projection and name the product formed by the oxidation of D-gulose.
Short Answer
- Glucose has six carbon atoms, it is classified as a hexose monosaccharide.
a)
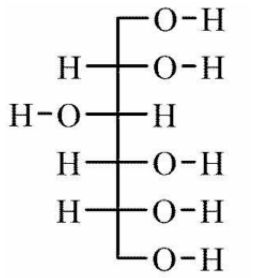
b)
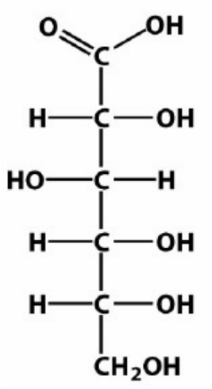




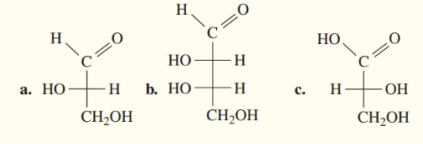
 b.
b. 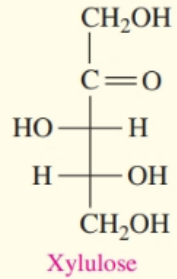
 d.
d.