Chapter 13: Q. 13.32 (page 450)
What are the kind and number of atoms in the ring portion of the Haworth structure of fructose?
Short Answer
In the cyclic shape of fructose, there are carbon atoms and an oxygen atom
Learning Materials
Features
Discover
Chapter 13: Q. 13.32 (page 450)
What are the kind and number of atoms in the ring portion of the Haworth structure of fructose?
In the cyclic shape of fructose, there are carbon atoms and an oxygen atom
All the tools & learning materials you need for study success - in one app.
Get started for free
D-Sorbitol, a sweetener found in seaweed and berries, contains only hydroxyl functional groups. When D-sorbitol is oxidized, it forms D-glucose. Draw the Fischer projection for D-sorbitol.
Indicate whether each disaccharide in the following figure is a reducing sugar or not.
a.
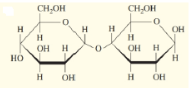
b.

Indicate whether each pair of Fischer projections represents enantiomers or identical structures.
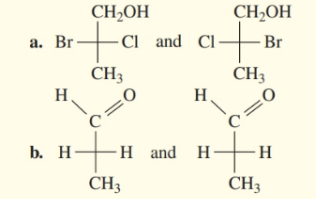
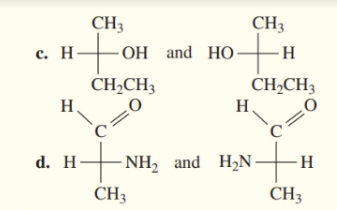


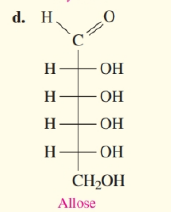
Draw the Fischer projection for the other enantiomer of a to d in problem 13.22.
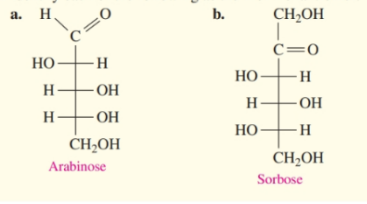
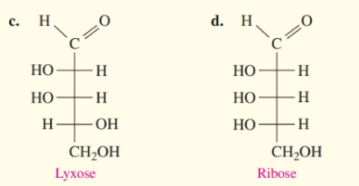
What do you think about this solution?
We value your feedback to improve our textbook solutions.