Chapter 4: Q. 4.69 (page 127)
Fill in the following blanks using higher or lower, more or less: Sr has a _____ionization energy and _______metallic character than Sb.
Short Answer
Sr has a lower ionization energy and more metallic character than Sb.
Learning Materials
Features
Discover
Chapter 4: Q. 4.69 (page 127)
Fill in the following blanks using higher or lower, more or less: Sr has a _____ionization energy and _______metallic character than Sb.
Sr has a lower ionization energy and more metallic character than Sb.
All the tools & learning materials you need for study success - in one app.
Get started for free
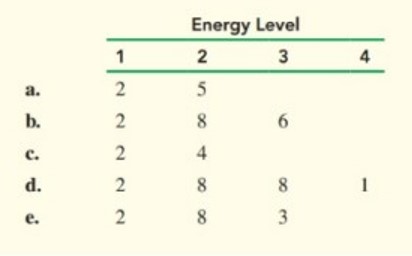
Identify the elements that have following electron arrangements:
There are five naturally occurring isotopes of zinc:,,,, and . None of these isotopes has the atomic mass of listed for zinc on the periodic table.
Explain.
Write the atomic symbol for the isotope with each of the following characteristics:
a. An oxygen atom with neutrons
b. protons and neutrons
c. electrons and 28 neutrons
d. A mass number of and neutrons
e. A nickel atom with neutrons
Place the elements in each set in order of increasing ionization energy.
a. O, N, C
b. S, P, Cl
c. As, P, N
d. Al, Si, P
Write the electron arrangement for each of the following elements: (Example: sodium )
a. Carbon
b. Argon
c. Potassium
d. Silicon
e. Helium
f. Nitrogen
What do you think about this solution?
We value your feedback to improve our textbook solutions.