Chapter 4: Q. 4.41 (page 115)
Copper consists of two isotopes, and .If the atomic
mass for copper on the periodic table is , are there more
atoms of and in a sample of copper?
Short Answer
There are more atoms of in a sample of copper.
Learning Materials
Features
Discover
Chapter 4: Q. 4.41 (page 115)
Copper consists of two isotopes, and .If the atomic
mass for copper on the periodic table is , are there more
atoms of and in a sample of copper?
There are more atoms of in a sample of copper.
All the tools & learning materials you need for study success - in one app.
Get started for free
Is each of the following statements true or false?
a. A proton and an electron have opposite charges.
b. The nucleus contains most of the mass of an atom.
c. Electrons repel each other.
d. A proton is attracted to a neutron.
What is the group number and number of valence electrons for each of the following elements?
a. Potassium
b. Silicon
c. Neon
d. Aluminum
e. Barium
f. Bromine
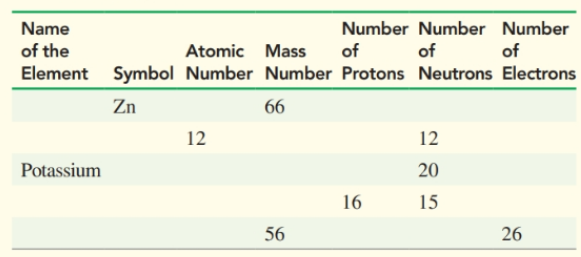
Complete the following table for atoms of essential elements in the body:
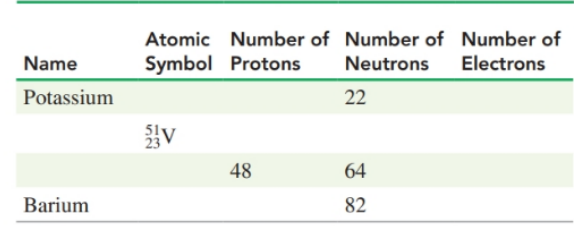
Complete the following table:
Give the symbol of the element described by each of the following:
a. the alkaline earth metal in Period
b. Group , Period
c. the noble gas in Period
d. the halogen in Period
e. Group , Period
What do you think about this solution?
We value your feedback to improve our textbook solutions.