Chapter 16: Q. 16.77 (page 581)
How would the lock-and-key model explain that sucrase hydrolyzes sucrose, but not lactose?
Short Answer
Sucrose conducts catalyzed since this fits the form of the active site in sucrase, however lactose doesn't quite.
Learning Materials
Features
Discover
Chapter 16: Q. 16.77 (page 581)
How would the lock-and-key model explain that sucrase hydrolyzes sucrose, but not lactose?
Sucrose conducts catalyzed since this fits the form of the active site in sucrase, however lactose doesn't quite.
All the tools & learning materials you need for study success - in one app.
Get started for free
Indicate whether each of the following would be a substrateor an enzyme
lactose
lipase
amylase
trypsin
pyruvate
transaminase
Aspartame, which is used in artificial sweeteners, contains the following dipeptide:
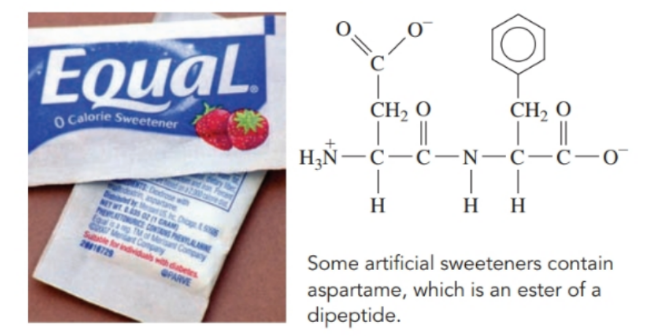
a. What are the amino acids in aspartame?
b. How would you name the dipeptide in aspartame?
How does the polarity of the R group in leucine compare to the R group in serine?
What is the reactant for each of the following enzymes?
a. peptidase
b. cellulase
c. lactase
Indicate whether each of the following describes a competitive or a noncompetitive enzyme inhibitor:
a. The inhibitor has a structure similar to the substrate.
b. The effect of the inhibitor cannot be reversed by adding more substrate.
c. The inhibitor competes with the substrate for the active site,
d. The structure of the inhibitor is not similar to the substrate.
e. The addition of more substrate reverses the inhibition.
What do you think about this solution?
We value your feedback to improve our textbook solutions.