Chapter 16: Q. 16.72 (page 581)
Maltase is an enzyme that hydrolyzes maltose to two glucose molecules.
What are the reactants and products of the reaction?
Draw an energy diagram for the reaction with and without maltase.
How does maltase make the reaction go faster?
Short Answer
Maltase is an enzyme that catalyses the hydrolysis of maltose:
Part a) Reactants and products of the reaction
part b)
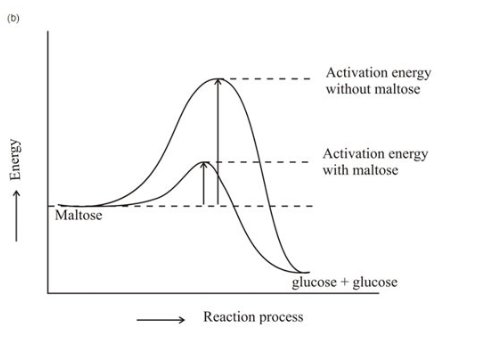
Part c) As the activation energy of the reaction falls, additional reactants join the reaction, and the process accelerates.



