Chapter 12: Q. 12.6 (page 405)
Give the common name for each of the following
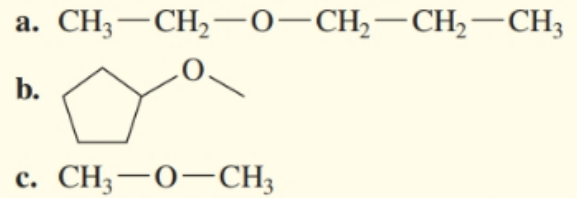
Short Answer
(a) The name of the compound is ethyl propyl ether.
(b) The name of the compound is cyclopentyl methyl ether.
(c) The name of the structure is dimethyl ether.
Learning Materials
Features
Discover
Chapter 12: Q. 12.6 (page 405)
Give the common name for each of the following
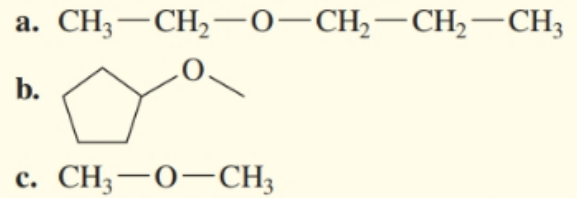
(a) The name of the compound is ethyl propyl ether.
(b) The name of the compound is cyclopentyl methyl ether.
(c) The name of the structure is dimethyl ether.
All the tools & learning materials you need for study success - in one app.
Get started for free
A compound with the formula is synthesized from and oxidizes easily to give a carboxylic acid. Draw the condensed structural formula and give the IUPAC name for the compound.
One of the components of gasoline is octane, , which has a density of . The combustion of mole of octane provides . A hybrid car has a fuel tank with a capacity of and a gas mileage of
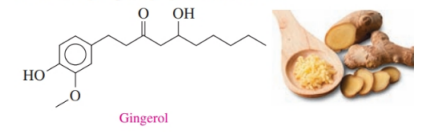
Gingerol is a pungent compound found in ginger. Identify the functional groups in gingerol.
In a teaspoon of a common liquid antacid, there are
of and of . A solution, which is similar to stomach acid, is used to
neutralize of the liquid antacid.
a.Write the balanced chemical equation for the neutralization
of and
b.Write the balanced chemical equation for the neutralization
of and
c. What is the pH of the solution?
d. How many milliliters of the solution are needed to
neutralize the ?
e. How many milliliters of the solution are needed to
neutralize the ?
Classify each of the following alcohols as primary (), secondary () , or tertiary ():()
a.
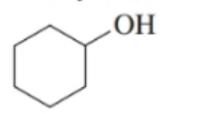
b.
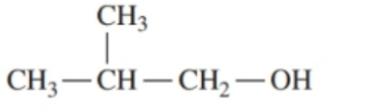
c.
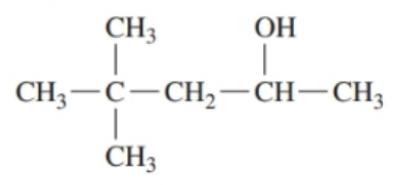
d.
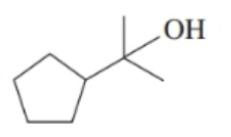
What do you think about this solution?
We value your feedback to improve our textbook solutions.