Chapter 12: Q. 12.50 (page 426)
Which compound in each pair would be more soluble in water? Explain. ()
a. or
b. or
c. or
Short Answer
(a) The will be more soluble in water
(b) The will be more soluble in water
(c) The will be more soluble in water
Learning Materials
Features
Discover
Chapter 12: Q. 12.50 (page 426)
Which compound in each pair would be more soluble in water? Explain. ()
a. or
b. or
c. or
(a) The will be more soluble in water
(b) The will be more soluble in water
(c) The will be more soluble in water
All the tools & learning materials you need for study success - in one app.
Get started for free
A compound with the formula is synthesized from and oxidizes easily to give a carboxylic acid. Draw the condensed structural formula and give the IUPAC name for the compound.
Draw the condensed structural formulas and give the IUPAC name for all the alcohols that have the formula .
Butyraldehyde is a clear liquid solvent with an unpleasant odor. It has a low boiling point and is highly flammable. Butyraldehyde has a density of . The unpleasant odor of old gym socks is due to butyraldehyde.
Classify each of the following alcohols as primary () , secondary () , or tertiary () : ()
a.
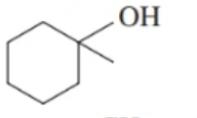
b.
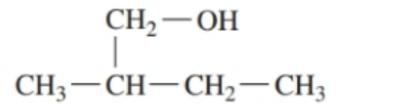
c.
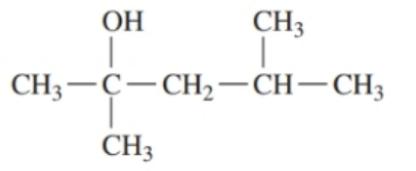
d.
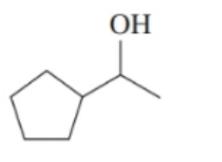
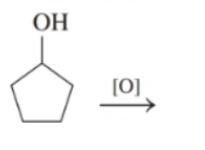
Draw the condensed structural or line-angle formula for the alkene, aldehyde, or ketone product of each of the following reactions: (12.4)
a.
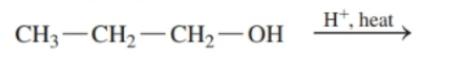
b.
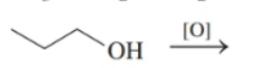
c.
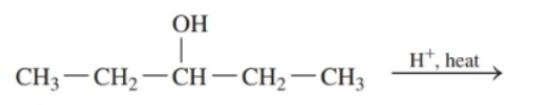
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.