Chapter 10: Q.10.51 (page 350)
What is the molarity of a solution of HCL if 5ml of the HCL solution is titrated with 28.6ml of a 0.145 M NaOH solution?
Short Answer
The molarity of a solution of HCL is M.
Learning Materials
Features
Discover
Chapter 10: Q.10.51 (page 350)
What is the molarity of a solution of HCL if 5ml of the HCL solution is titrated with 28.6ml of a 0.145 M NaOH solution?
The molarity of a solution of HCL is M.
All the tools & learning materials you need for study success - in one app.
Get started for free
If of a solution is required to titrate of a solution of , what is the molarity of the solution?
Identify the reactant that is a Brønsted-Lowry acid and the reactant that is a Brønsted-Lowry base in each of the following:
a.
b.
Use Le Châtelier's principle to predict whether each of the following changes causes the system to shift in the direction of products or reactants:
a. adding more
b. removing some
c. removing some
d. adding more
Calculate the pH of each solution given the follow in:
a.
b.
c.
d.
e.
f.
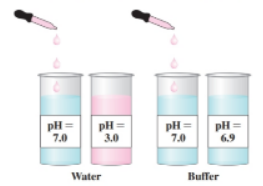
Adding a few drops of a strong acid io water will lower the phi appreciably. However, adding the same number of drags to a buffer does not appreciably alter the ph. Why? (10.8)
What do you think about this solution?
We value your feedback to improve our textbook solutions.