Chapter 10: Q. 10.27 (page 340)
In an acidic solution, how does the concentration of compare to the concentration of ?
Short Answer
The concentration is greater than .
Learning Materials
Features
Discover
Chapter 10: Q. 10.27 (page 340)
In an acidic solution, how does the concentration of compare to the concentration of ?
The concentration is greater than .
All the tools & learning materials you need for study success - in one app.
Get started for free
Identify the stronger acid in each of the following pairs: (10.3)
a. HF or HCN
b.
c.
d.
Identify each of the following as an acid, base, or salt. and give its name: (10.1)
a.
b.
c.
d.
e.
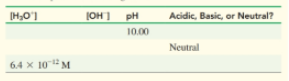
Complete the following table :
Sometimes, during stress or trauma, a person can start to hyperventilate. Then the person might breathe into a paper bag to avoid fainting. (10.8)
a. What changes occur in the blood during hyperventilation?
h. How does breathing into a paper bag help return blood pH to normal??
Which of the following represents a buffer system? Explain.
a. and
b. and
c. and
d. and
What do you think about this solution?
We value your feedback to improve our textbook solutions.