Chapter 9: Q9.1-2CC. (page 169)
If the following redox reaction occurred, which compound would be oxidized? Reduced?
Short Answer
In the given case, will get oxidized and will get reduced.
Learning Materials
Features
Discover
Chapter 9: Q9.1-2CC. (page 169)
If the following redox reaction occurred, which compound would be oxidized? Reduced?
In the given case, will get oxidized and will get reduced.
All the tools & learning materials you need for study success - in one app.
Get started for free
Membranes must be filled with fluid to function properly(as you learned in Concept 7.1). How does the operation of the electron transport chain support that assertion?
During redox reaction in glycolysis (see step 6 in figure 9.9), which molecule act as oxidizing agent? The reducing agent?
The conversion shown in Figure 9.10 and step 4 of figure 9.12 are catalyzed by a large multienzyme complex. What similarities are there in the reactions that occur in these two cases?
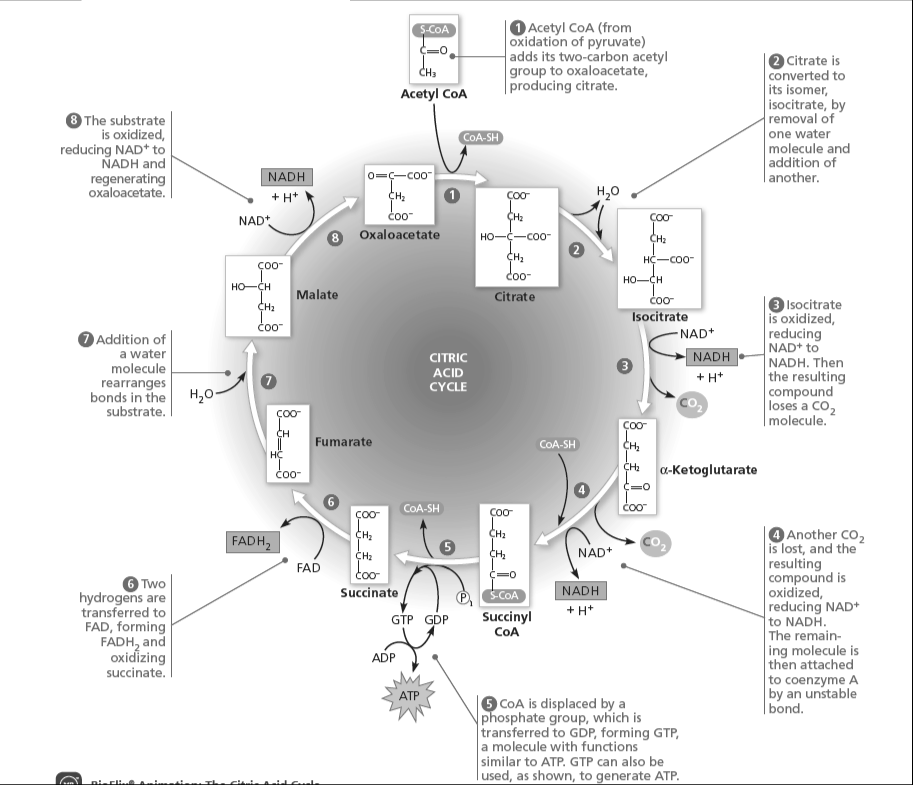
Compare and contrast aerobic and anaerobic respiration, including the processes involved.
What processes in your cells produce CO2 that you exhale?
What do you think about this solution?
We value your feedback to improve our textbook solutions.