Chapter 4: Q4.2-1CC (page 62)
(a) Draw a structural formula for C2H4.
(b) Draw the trans isomer of C2H2Cl2.
Learning Materials
Features
Discover
Chapter 4: Q4.2-1CC (page 62)
(a) Draw a structural formula for C2H4.
(b) Draw the trans isomer of C2H2Cl2.
All the tools & learning materials you need for study success - in one app.
Get started for free
The earlier published experiment carried out by Miller did not include H2S in the reactants (see Figure 4.2). Which compounds shown in the data table can be made in the H2S experiment but could not be made in the earlier investigation?
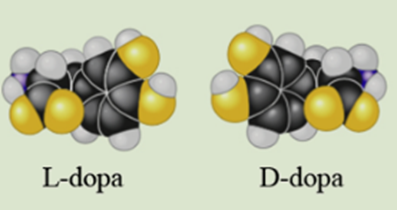
In 1918, an epidemic of sleeping sickness caused an unusual rigid paralysis in some survivors, similar to symptoms of advanced Parkinson’s disease. Years later, L-dopa (below, left), a chemical used to treat Parkinson’s disease, was given to some of these patients. L-dopa as remarkably effective at eliminating the paralysis, at least temporarily. However, its enantiomer, D-dopa (right), was subsequently shown to have no effect at all, as is the case for Parkinson’s disease. In a short essay (100-150 words), discuss how the effectiveness of one enantiomer and not the other illustrates the theme of structure and function.
Which of the molecules shown in question 5 has an asymmetric carbon? Which carbon is asymmetric?
Why was Wohler astonished to find he had made urea?
Which two pairs of molecules in Figure 4.5 are isomers? For each pair, identify the type of isomer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.