Chapter 6: Q. 2 (page 175)
Figure 6.10 If no activation energy were required to break down sucrose (table sugar), would you be able to store it in a sugar bowl?
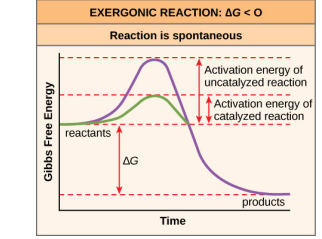
Short Answer
No, Sucrose can not be stored it in a sugar bowl.
Learning Materials
Features
Discover
Chapter 6: Q. 2 (page 175)
Figure 6.10 If no activation energy were required to break down sucrose (table sugar), would you be able to store it in a sugar bowl?

No, Sucrose can not be stored it in a sugar bowl.
All the tools & learning materials you need for study success - in one app.
Get started for free
An allosteric inhibitor does which of the following?
The energy released by the hydrolysis of ATP is____
Imagine an elaborate ant farm with tunnels and passageways through the sand where ants live in a large community. Now imagine that an earthquake shook the ground and demolished the ant farm. In which of these two scenarios, before or after the earthquake, was the ant farm system in a state of higher or lower entropy?
DNA replication involves unwinding two strands of parent DNA, copying each strand to synthesize complementary strands, and releasing the parent and daughter DNA. Which of the following accurately describes this process?
a. This is an anabolic process.
b. This is a catabolic process.
c. This is both anabolic and catabolic.
d. This is a metabolic process but is neither anabolic nor catabolic.
Consider a pendulum swinging. Which type(s) of energy is/are associated with the pendulum in the following instances:
i. The moment at which it completes one cycle, just before it begins to fall back towards the other end.
ii. The moment that it is in the middle between the two ends, and
iii. Just before it reaches the end of one cycle (just before instant I.).
a. i. potential and kinetic, ii. potential and kinetic, iii. kinetic
b. i. potential, ii. potential and kinetic, iii. potential and kinetic
c. i. potential, ii. kinetic, iii. potential and kinetic
d. i. potential and kinetic, ii. kinetic iii. kinetic
What do you think about this solution?
We value your feedback to improve our textbook solutions.